Analysis of the effect of resistance increase on the capacity fade of Li-ion batteries
This post is a review of “Analysis of the effect of resistance increase on the capacity fade of lithium ion batteries” by Mandli, Kaushik, Patil, Naha, Hariharan, Kolake, Han, and Choi.
Simple vs. complicated cell models
The authors point out that various research groups suggested many physical cell capacity fade models which sometimes assume very different degradation mechanisms, SEI growth, loss of active material, and Lithium plating:
Ramadass et al [1] have formulated a model attributing the capacity fade to the loss of Lithium inventory on the negative electrode. Safari et al [2] model assumes that the solvent decomposition reaction at the anode leading to SEI growth is the source of capacity fade. The models of Arora et al [3] and Tang et al [4] have incorporated lithium plating as a reason for capacity fade. Dubarry et al [5] have assumed an exponential loss of active material to model the nonlinear capacity fade behavior. Yang et al [6] have attributed the nonlinear capacity fade occurring in lithium ion batteries to an exponential increase in lithium plating.
Despite appearing contradictory on the surface, all or most of these results might be largely correct: different research groups have used different protocols to cycle cells of various form and chemistry, so the leading degradation mechanisms in their experiments might genuinely be different.
From the above observation, the authors conclude that battery management systems should implement simple semi-empirical models instead of physics-based models:
The complexity of these detailed models makes them largely unattractive for developing estimation and optimization algorithms that can be implemented in microcontrollers. [...] Many assumptions, together with the many parameters, present in detailed battery degradation models complicate an understanding of the major influencing factors of battery degradation.
The development of estimation and optimization algorithms for battery life management that are suitable for microcontroller implementation requires simple semi-empirical models that can capture degradation accurately.
I agree that this is not necessary to use physics-based models in the BMS: for field operation (rather than research), we should always choose the most accurate model available, regardless of whether this model is physics-based or semi-empirical. However, the model used in the BMS should also be able to correctly attribute the changes in the cell sensor output to shifts in different cell parameters to estimate the risk of cell failure. Therefore, I'm not sure that the cell model used in the BMS should be too simple: such a model might estimate some cell parameters inadequately when it "stretches itself" to explain some unusual cell sensor outputs. In other words, that would be an underfitted model.
The rise of cell internal resistance can explain the capacity fade "knee"
The authors demonstrate that under some assumptions, the "knee" in the typical cell capacity fade curve (when after some number of cycles the cell enters a different mode with a higher capacity fade rate per each cycle) can be solely explained by the increase in cell internal resistance.
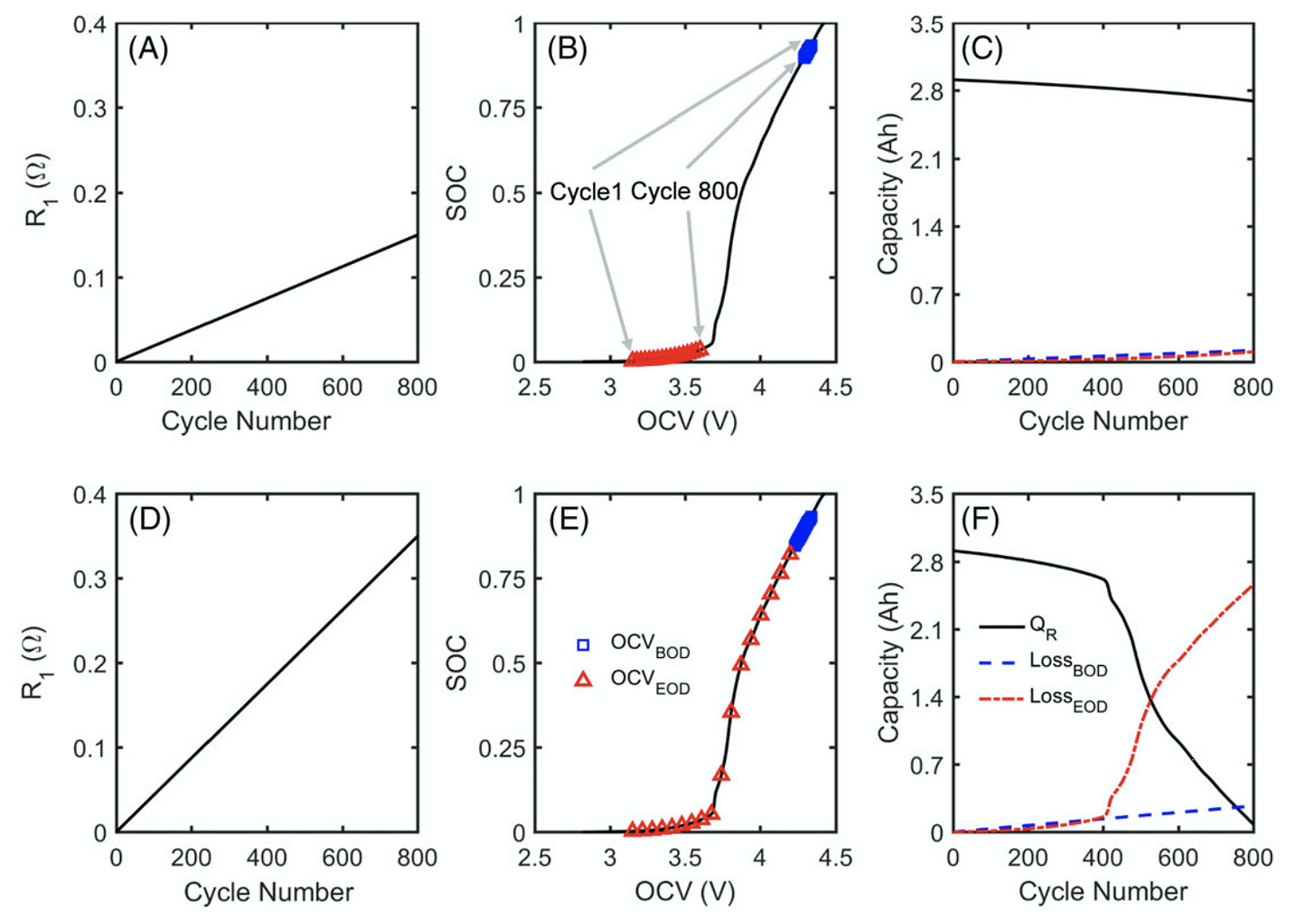
You can see the knee on the lower-right chart, the black line which models the cell capacity:
In the figure above, R_1 models the increase of the cell internal resistance (but not diffusion resistance) relative to the baseline level which is assumed to be 0.15 Ohm. Blue squares and red triangles designate the points on the open-circuit voltage curve where the cell charging (OCV_BOD = "beginning of discharge" = end of charge) and discharging (OCV_EOD = "end of discharge") should stop, respectively, assuming the CCCV charging protocol with cutoff C/10 rate in the constant voltage phase, and 1C constant-current discharging protocol.
The model shows that if cell internal resistance raises by 100% from 0.15 Ohm to 0.15+0.15 = 0.3 Ohm (see the top row of charts) then this change can only explain a slight decrease in cell capacity. But if cell internal resistance increases by 233% to 0.15+0.35=0.5 Ohm (see the bottom row of charts) then this change can explain accelerated capacity fade. Cell discharge stops when the open-circuit voltage is still on the plateau.
Here is a real cycling experiment:
The meaning of blue squares and red triangles is the same as in the figure above. The rightmost chart demonstrates the results of the experiment: Q_Exp means "experimental", i. e. the measured capacity, and Q_R is the predicted capacity assuming the increase of cell resistance is the only reason for the capacity fade.
It's important to note that this cell was cycled at 45 °C, probably to induce severe SEI growth to make the cell resistance increase as much as the model above "requires" (hundreds of percent). I think that under normal conditions, cell internal resistance should not increase by more than 20-30% (e. g., demonstrated in this study).
Also, the authors characterised the cell discharge capacity at 1C rate, which is not typical in experiments but was needed to demonstrate the knee effect on a real cell.
One thing that seems questionable to me in explaining this experimental result by the model proposed above is that the gap between the measured capacity and the predicted capacity increases until about the 600th cycle (which would be expected, meaning that there are some other factors contributing to the capacity fade other than the rise in cell resistance, e. g., the loss of Lithium material) and decreases after the 600th cycle meaning that some of the capacity fade mechanisms are reversed, which is impossible in this experiment.
Discussion
This model is at odds with the theory that cell capacity fade accelerates when Lithium deposition becomes irreversible. However, I'm inclined to think that both theories are right under some circumstances. The internal resistance can play a significant role only in energy cells, that the authors themselves point out:
This contribution is likely to be highly significant in energy cells compared with power cells where initial diffusional resistances are low.
Albeit the authors didn't consider diffusion resistance in the model, it's more likely that in reality, diffusion resistance plays an even bigger role in limiting the cell's 1C discharge capacity when the SEI layer grows.
Even in energy cells, to be able to explain the knee on the capacity fade curve, the internal resistance should increase by hundreds of percent which is only possible when cells are cycled at a very high temperature. However, in different experiments in literature, we see that the capacity fade curve can have a knee even when the cell is cycled at the standard temperature.
In general, the fact that the rise in cell internal resistance can contribute to the observed cell capacity fade reminds me that cell data mining, analysis, and modelling are greatly complicated by factors that are not essential to cell electrochemistry.
References
[1] Development of first principles capacity fade model for Li‐ion cells (2004)
[2] Multimodal physics‐based aging model for life prediction of Li‐ion batteries (2008)
[4] Two-Dimensional Modeling of Lithium Deposition during Cell Charging (2009)
[5] Synthesize battery degradation modes via a diagnostic and prognostic model (2012)